
CER Groupe conducts toxicology, safety and clinical studies for veterinary medicinal products, fully aligned with VICH and CVMP requirements.
Are you looking for a trusted GLP partner to generate robust non-clinical safety data for your product?
CER Groupe is GLP-certified by the Belgian Competent Authority (Sciensano) since 1993, with a dedicated GLP toxicology scope since 2010.
Studies performed
GLP-Certifed since 1993
Week to introduce a study
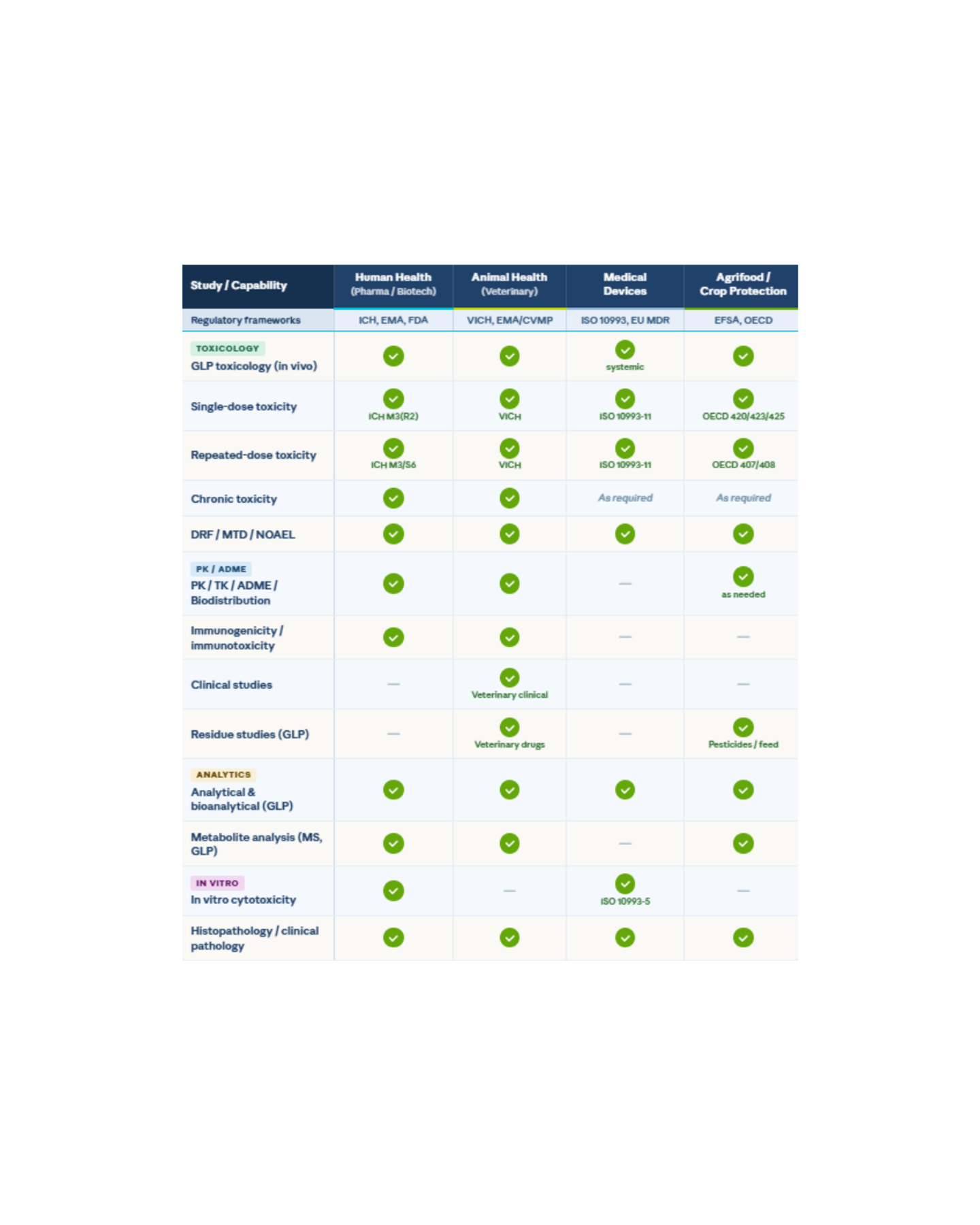
CER Groupe delivers Good Laboratory Practice (GLP)-compliant toxicology and safety studies. All in vivo toxicology studies are designed according to ICH, VICH, OECD and ISO guidance, with a strong focus on scientific relevance, animal welfare and regulatory acceptance. Study duration, dose levels, group size and endpoints are defined based on the intended clinical or regulatory use, ensuring data are both fit-for-purpose and inspection-ready.
Human medicinal products
Veterinary medicinal products
Novel Food, food ingredients, feed additives, pesticides, other crop solutions and bioactive substances for EFSA submissions
Our services cover the full workflow, from study design to final archiving, combining:
We support you across the full non-clinical lifecycle:
Study design → protocol development → execution → QA & reporting → archiving
The table below summarizes all study types, combining transversal evaluations with item- and host-specific studies.

CER Groupe conducts toxicology, safety and clinical studies for veterinary medicinal products, fully aligned with VICH and CVMP requirements.

CER Groupe conducts toxicology, safety and PK/TK studies for human and animal health products, fully aligned with ICH guidelines.
CER Groupe supports medical device manufacturers with ISO 10993 biocompatibility studies for Class II and III devices under MDR.

CER Groupe supports agrifood, feed and crop developers with tailored toxicology and analytical studies, ensuring safety and regulatory acceptance.
In addition to animal studies, CER Groupe provides standalone GLP analytical services, supporting a wide range of regulatory contexts.

Applicable to

Technologies
Fully compliant with OECD GLP and regulatory guidance for metabolites in human, veterinary and agrifood contexts.
CER Groupe integrates comprehensive monitoring techniques adapted to your study objectives - ensuring early detection of adverse effects and reliable toxicological interpretation.
These endpoints contribute to early detection of adverse effects, improved toxicological interpretation, and stronger benefit-risk assessment.
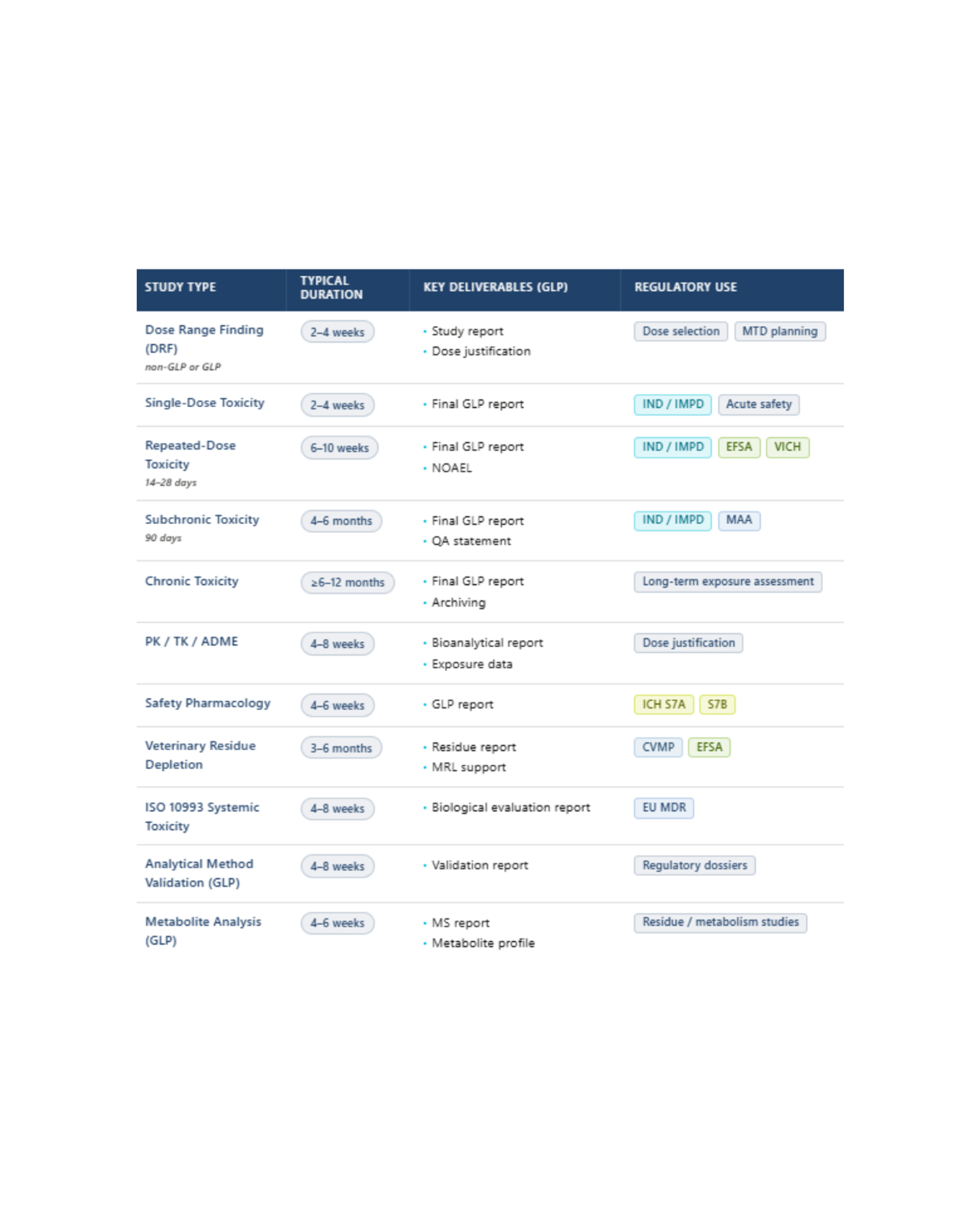
Indicative timelines may vary depending on product type, study design, species and regulatory requirements
8–12 weeks from study agreement to first dosing (GLP) - shorter timelines available for analytical-only or non-GLP exploratory studies.
